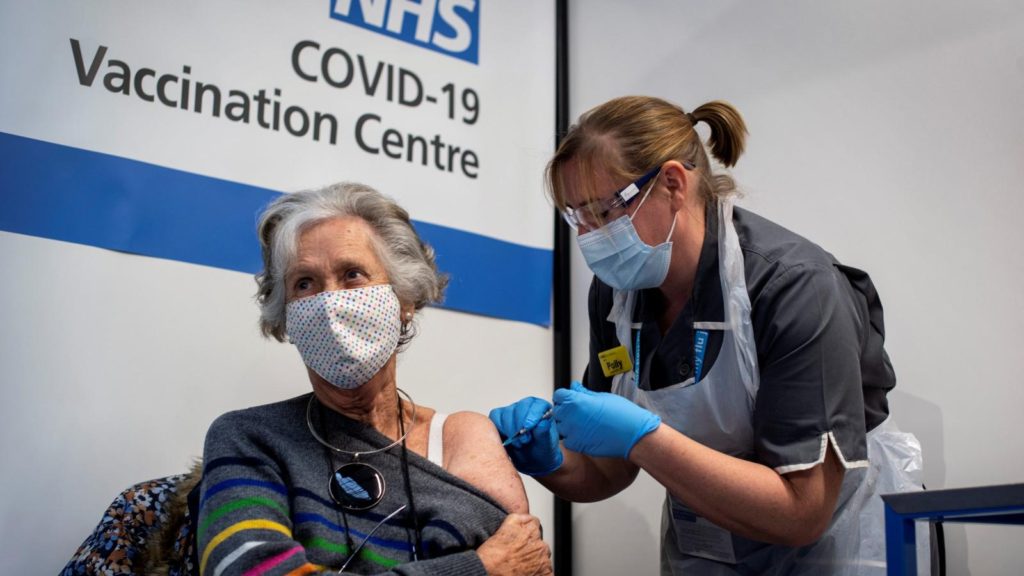
COVID-19 Vaccine Distribution Begins
Back in March, researchers had to quickly develop a vaccine that would mitigate the COVID-19 virus. Multiple pharmaceutical companies started developing a vaccine, which gave people a sign of hope to gain back normalcy. Creating a vaccine takes a large amount of research and a couple of years. The fastest vaccine development was the mumps vaccine which took four years. However, scientists have been researching hundreds of coronaviruses for over fifty years and understand this type of virus’s genome and structure. Under normal circumstances, a regular vaccine would normally take a decade or more to develop. However, as previously mentioned, the collection of former data as well as worldwide collaboration in the scientific community were important factors in developing the COVID-19 vaccine, and therefore making it a quicker process.
In other countries, there were several organizations that were already in the process of developing a vaccine. The United States has approved the COVID-19 vaccine from two different companies: Pfizer-BioNTech and Moderna. The countries that the vaccine was developed in were Australia, Germany, and the United Kingdom. Companies within these countries have private pharmaceutical firms that collaborate with public health research and university labs. The government is able to provide funds through public health agencies, which were the Department of Health and Human Services (HHS), Centers for Disease Control and Prevention (CDC), the National Institutes of Health (NIH), Food and Drug Administration (FDA), and the Department of Defense in the United States. Assistance was also gained from international institutions such as the World Health Organization (WHO) and Coalition for Epidemic Preparedness Innovations (CEPI) working for international global use of the vaccine.
The vaccine process went through several different stages such as research, preclinical preparation, clinical trials, approval, manufacturing, and distribution. Typically, vaccines create antibodies within your body to help fight the virus by giving antigens, weakened virus molecules, for your immune system to understand how to combat it. The type of vaccine that Pfizer and Moderna created uses the genetic material, like DNA and RNA, of the virus to create antibodies. The most popular vaccine is the mRNA vaccine but there is also the protein subunit vaccine and vector vaccine. Most COVID-19 vaccines require two doses of the shot. In the United States, there are two shots that need to be taken. It is important to note that a person cannot get one shot from one company, such as Pfizer, and take a second shot from another like Moderna. The two shots must come from the same company.
The first person to get the COVID-19 vaccine was an elderly woman by the name of Margaret Keenan in the United Kingdom. She had used the vaccine from the Pfizer lab, which was the first dosage used out of the 800,000 shots that were meant to be distributed to the public. In the United States, the first person to get the Pfizer-BioNTech vaccine was a frontline worker named Sandra Lindsay in December. Now, South Florida has received vaccines from the federal government to distribute within counties. The main priority is to vaccinate the elderly (age 65 or older) and essential workers with the general public following. To set up an appointment for the COVID-19 vaccine, if any of your family fall under those requirements, visit Miami’s COVID Vaccine site here: https://www.miamidade.gov/global/initiatives/coronavirus/vaccine/home.page or talk with your healthcare provider.
You may be interested
Rocking The Halls of Ferguson
Sofia Morales - Apr 23, 2024Out of all the events that Ferguson hosts throughout the year, one of the most anticipated is the seasonal rock concerts. Students are…

Ms. Sol and Her Knack for Cats
Emi Martin - Apr 23, 2024Every school day, Ms. Sol enters the classroom to teach English and Literature, interacting with ninth and twelfth-grade students throughout the day. As…

Ferguson’s Athletic Director: Shanower
Anela Armenis - Apr 19, 2024Gregory Shanower, the athletic director of Ferguson Falcons, has been teaching for over 21 years. He has faced numerous challenges throughout his career,…